Mass Number Atomic Number
Atomic number and mass number are always whole numbers because they are obtained by counting whole objects (protons, neutrons, and electrons). The sum of the mass number and the atomic number for an atom (A-Z) corresponds to the total number of subatomic particles present in the atom. Atomic Number & Mass Number Properties of Matter Chemistry FuseSchoolHow do we tell elements apart from each other? Find out in this video from the Pr. Mass number is an integer (whole number) equal to the sum of the number of protons and neutrons of an atomic nucleus. In other words, it is the sum of the number of nucleons in an atom. Mass number is often denoted using a capital letter A. Contrast this with the atomic number, which is simply the number of protons. The mass of the atom of a particular isotope relative to hydrogen 1 (or to one twelfth the mass of carbon 12), generally very close to the whole number represented by the sum of the protons and neutrons in the atomic nucleus of the isotope; it is not to be confused with the atomic weight of an element, which may include a number of isotopes in natural proportion. The main difference between atomic number and mass number is that the atomic number indicates the number of protons present in an atom whereas, the mass number indicates the sum of the number of protons and the number neutrons present in an atom.
Atomic Number
The total number of protons present in the nucleus of an atom is known as its atomic number. For example, Nitrogen has seven protons, so its atomic number is seven. It is denoted by the letter Z of English.
It is a number assigned to a particular element based on the number of protons present in its atoms. All the atoms of an element contain the same number of protons. Besides this, it is a unique number as two different elements cannot have the same number of protons, e.g., for example, oxygen has eight protons and no other element can have eight protons. So, atomic number acts as the identity of an element.
Besides this, atomic number defines the characteristics or properties of an element as in a neutral atom, it is equal to the number of electrons, which determine the valency (reactive capacity) of an atom. The reactive capacity decides the properties of elements. So, it is also the characteristic property of an element.
Furthermore, the atomic number is always a whole number as the number of protons is fixed in an atom. The number of electrons may change as they take part in chemical reactions where an atom may lose or gain electrons.
Atomic Mass
It is the mass of a single atom of an element, which includes the total number of protons and neutrons present in the nucleus of an atom. It does not include electrons as their weight is negligible as compared to protons and neutrons that have a significant amount of mass. We can say that electrons do not contribute to the mass number of an atom as their mass is negligible as compared to the mass of a proton or a neutron. So, electrons are considered weightless.
Atomic mass is also referred to as the mass number. So, we can say that the sum of all protons and neutrons present in the nucleus of an atom is called its mass number. The protons and neutrons are collectively known as nucleons as they are contained in the nucleus. So, we can say that the atomic mass or mass number is equal to the total number of nucleons present in the atom of an element.
How is atomic mass measured?
The protons and neutrons that contribute to the atomic mass are present in the nucleus, which is present at the centre of the atom and its size is very small as compared to the atom. So, the mass of atoms is so small that it was difficult for the scientist to measure and represent its mass. For example, the atomic mass of a hydrogen atom is 1.67 x 10-27 kg. This way of representing atomic mass was not easy or appropriate, so to simplify the method or to assign an easy atomic mass figure, the concept of relative atomic mass was introduced by the scientists and a new unit called atomic mass unit (amu) was introduced to calculate the atomic mass of an atom.
What is the atomic mass unit (amu)?
The atomic mass unit is the unit to measure and represent the atomic mass. It is equal to one-twelfth (1/12) of the mass of a carbon-12 atom. For example, if we divide C 12 atom into 12 equal pieces and take one piece then we can say that mass of this one piece is equal to 1 atomic mass unit, which is used as a standard to calculate the mass of any other atom. So, 1 amu = 1/12th x mass of C-12. There are 12 nucleons (protons + neutrons) in the nucleus of Carbon. So, when we divide it into twelve parts, 1 amu is nearly equal to the 1 proton. As of now amu is simply known as unified atomic mass and is written as 'u'.
So, Atomic mass of an element = Mass number of one atom of the element (protons + neutrons) / (1/12) x mass of carbon
For example, atomic mass of oxygen in amu = 16 (8 proton + 8 neutron) / 1/12x 12 = 16 amu
So, we can say that the atomic mass of an element is the number of times its atom is heavier than 1/12th of the mass of an atom of C-12, e.g., the atomic mass of Mg is 24 u. It means the atomic mass or mass of one atom of Magnesium is 24 times more than the 1/12 of the atomic mass of carbon 12.
From the above relation between the mass number and protons and neutrons. We can calculate the number of neutrons if we have the mass number and number of protons of an element. Similarly, the number of protons can be calculated if the mass number and number of neurons are given.
For example, Atomic mass of Aluminium = 27

No. of protons are given = 13
So, No. of neutrons = 27 - 13 (atomic mass - number of protons) = 14
Similarly, no of protons = 27 - 14 (atomic mass - number of neutrons) = 13
Similarly, if the atomic number (Z) and atomic mass (A) of an element are given, we can find out number of protons, neutrons and electrons in the given element. For example,
The atomic number of Aluminium is 13 and its atomic mass or mass number is 27.
In this case,
- of protons = 13 as an atomic number is equal to the number of protons.
- The number of neutrons = atomic mass number - no. of protons (27 - 13) = 14 neutrons.
- The number of electrons = 13 as the number of electrons = number of protons in a neutral atom, which is not in an ionic state.
Difference between Atomic Number and Atomic Mass
| Atomic Number | Atomic Mass |
|---|---|
| It represents the total number of protons in the nucleus. | It represents the protons and neutrons present in the nucleus. |
| It does not depend on neutrons. | It is equal to the sum of protons and neutrons. |
| It is represented by 'Z'. | It is represented by 'A'. |
| It is the identity of an element. It is used in the classification and identification of elements. | It is not the identity of an element. It cannot be used to identify or define an element. |
| It is same for isotopes (different forms of an element). | It is different for isotopes. So, it is used to classify the different isotopes of an atom. |
| It is a number, which is used to place elements in the periodic table. | The unit to measure atomic mass is atomic mass unit (amu) or unified atomic mass (u). |
| The value of an atomic number of an element is always less than the value of the mass number. | Its value is always higher than the atomic number of the same element. |
How are the Atomic Number and the Mass Number of an element represented?
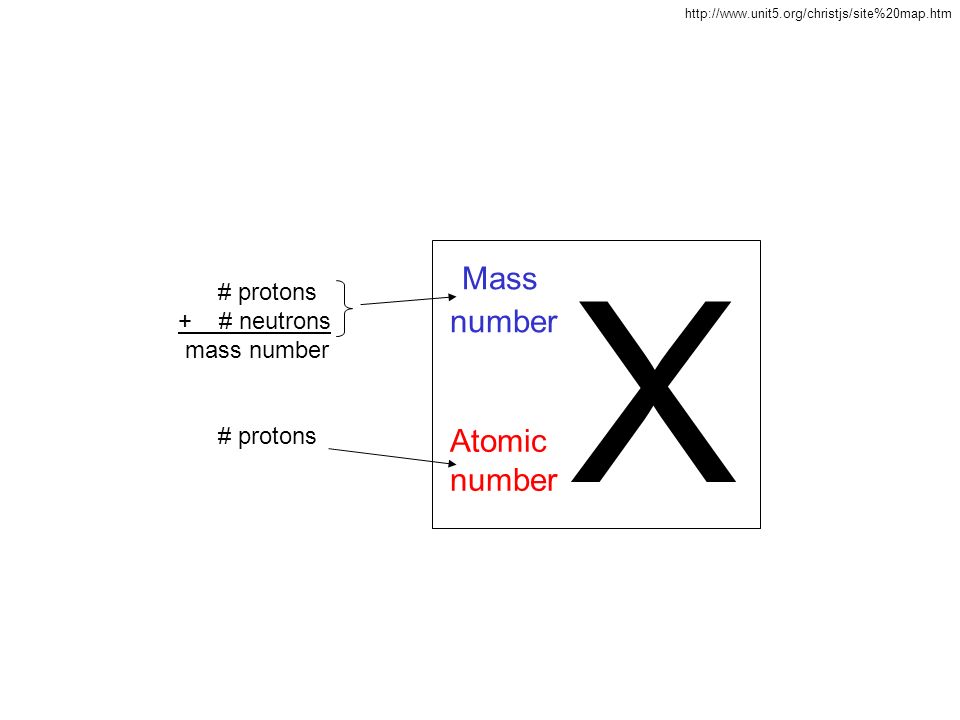
The atomic number of an element is usually denoted by the English letter Z, whereas the atomic mass is represented by the English alphabet 'A'. They are represented along with the symbol of the element as shown in the below image: Z comes at subscript and A is written at superscript to the left-hand side of the symbol of an element. For example, a single atom of carbon has 6 protons and 6 neutrons, so its atomic number is equal to 6 and mass number 12.
Similarly, a single atom of Aluminium has 13 protons and 14 neutrons, so its atomic number is 13 and mass number is 27 as shown in the below image.
The atomic number or proton number (symbol Z) of a chemical element is the number of protons found in the nucleus of every atom of that element. The atomic number uniquely identifies a chemical element. It is identical to the charge number of the nucleus. In an uncharged atom, the atomic number is also equal to the number of electrons.
The sum of the atomic number Z and the number of neutronsN gives the mass numberA of an atom. Since protons and neutrons have approximately the same mass (and the mass of the electrons is negligible for many purposes) and the mass defect of nucleon binding is always small compared to the nucleon mass, the atomic mass of any atom, when expressed in unified atomic mass units (making a quantity called the 'relative isotopic mass'), is within 1% of the whole number A.
Atoms with the same atomic number but different neutron numbers, and hence different mass numbers, are known as isotopes. A little more than three-quarters of naturally occurring elements exist as a mixture of isotopes (see monoisotopic elements), and the average isotopic mass of an isotopic mixture for an element (called the relative atomic mass) in a defined environment on Earth, determines the element's standard atomic weight. Historically, it was these atomic weights of elements (in comparison to hydrogen) that were the quantities measurable by chemists in the 19th century.
The conventional symbol Z comes from the German word Zahl meaning number, which, before the modern synthesis of ideas from chemistry and physics, merely denoted an element's numerical place in the periodic table, whose order is approximately, but not completely, consistent with the order of the elements by atomic weights. Only after 1915, with the suggestion and evidence that this Z number was also the nuclear charge and a physical characteristic of atoms, did the word Atomzahl (and its English equivalent atomic number) come into common use in this context.
History[edit]
The periodic table and a natural number for each element[edit]
Loosely speaking, the existence or construction of a periodic table of elements creates an ordering of the elements, and so they can be numbered in order.
Dmitri Mendeleev claimed that he arranged his first periodic tables (first published on March 6, 1869) in order of atomic weight ('Atomgewicht').[1] However, in consideration of the elements' observed chemical properties, he changed the order slightly and placed tellurium (atomic weight 127.6) ahead of iodine (atomic weight 126.9).[1][2] This placement is consistent with the modern practice of ordering the elements by proton number, Z, but that number was not known or suspected at the time.
A simple numbering based on periodic table position was never entirely satisfactory, however. Besides the case of iodine and tellurium, later several other pairs of elements (such as argon and potassium, cobalt and nickel) were known to have nearly identical or reversed atomic weights, thus requiring their placement in the periodic table to be determined by their chemical properties. However the gradual identification of more and more chemically similar lanthanide elements, whose atomic number was not obvious, led to inconsistency and uncertainty in the periodic numbering of elements at least from lutetium (element 71) onward (hafnium was not known at this time).
The Rutherford-Bohr model and van den Broek[edit]
In 1911, Ernest Rutherford gave a model of the atom in which a central nucleus held most of the atom's mass and a positive charge which, in units of the electron's charge, was to be approximately equal to half of the atom's atomic weight, expressed in numbers of hydrogen atoms. This central charge would thus be approximately half the atomic weight (though it was almost 25% different from the atomic number of gold (Z = 79, A = 197), the single element from which Rutherford made his guess). Nevertheless, in spite of Rutherford's estimation that gold had a central charge of about 100 (but was element Z = 79 on the periodic table), a month after Rutherford's paper appeared, Antonius van den Broek first formally suggested that the central charge and number of electrons in an atom was exactly equal to its place in the periodic table (also known as element number, atomic number, and symbolized Z). This proved eventually to be the case.
Moseley's 1913 experiment[edit]
The experimental position improved dramatically after research by Henry Moseley in 1913.[3] Moseley, after discussions with Bohr who was at the same lab (and who had used Van den Broek's hypothesis in his Bohr model of the atom), decided to test Van den Broek's and Bohr's hypothesis directly, by seeing if spectral lines emitted from excited atoms fitted the Bohr theory's postulation that the frequency of the spectral lines be proportional to the square of Z.
To do this, Moseley measured the wavelengths of the innermost photon transitions (K and L lines) produced by the elements from aluminum (Z = 13) to gold (Z = 79) used as a series of movable anodic targets inside an x-ray tube.[4] The square root of the frequency of these photons (x-rays) increased from one target to the next in an arithmetic progression. This led to the conclusion (Moseley's law) that the atomic number does closely correspond (with an offset of one unit for K-lines, in Moseley's work) to the calculated electric charge of the nucleus, i.e. the element number Z. Among other things, Moseley demonstrated that the lanthanide series (from lanthanum to lutetium inclusive) must have 15 members—no fewer and no more—which was far from obvious from known chemistry at that time.
Missing elements[edit]
After Moseley's death in 1915, the atomic numbers of all known elements from hydrogen to uranium (Z = 92) were examined by his method. There were seven elements (with Z < 92) which were not found and therefore identified as still undiscovered, corresponding to atomic numbers 43, 61, 72, 75, 85, 87 and 91.[5] From 1918 to 1947, all seven of these missing elements were discovered.[6] By this time, the first four transuranium elements had also been discovered, so that the periodic table was complete with no gaps as far as curium (Z = 96).
The proton and the idea of nuclear electrons[edit]
Mass Number Atomic Number Atomic Mass
In 1915, the reason for nuclear charge being quantized in units of Z, which were now recognized to be the same as the element number, was not understood. An old idea called Prout's hypothesis had postulated that the elements were all made of residues (or 'protyles') of the lightest element hydrogen, which in the Bohr-Rutherford model had a single electron and a nuclear charge of one. However, as early as 1907, Rutherford and Thomas Royds had shown that alpha particles, which had a charge of +2, were the nuclei of helium atoms, which had a mass four times that of hydrogen, not two times. If Prout's hypothesis were true, something had to be neutralizing some of the charge of the hydrogen nuclei present in the nuclei of heavier atoms.
In 1917, Rutherford succeeded in generating hydrogen nuclei from a nuclear reaction between alpha particles and nitrogen gas,[7] and believed he had proven Prout's law. He called the new heavy nuclear particles protons in 1920 (alternate names being proutons and protyles). It had been immediately apparent from the work of Moseley that the nuclei of heavy atoms have more than twice as much mass as would be expected from their being made of hydrogen nuclei, and thus there was required a hypothesis for the neutralization of the extra protons presumed present in all heavy nuclei. A helium nucleus was presumed to be composed of four protons plus two 'nuclear electrons' (electrons bound inside the nucleus) to cancel two of the charges. At the other end of the periodic table, a nucleus of gold with a mass 197 times that of hydrogen was thought to contain 118 nuclear electrons in the nucleus to give it a residual charge of +79, consistent with its atomic number.
The discovery of the neutron makes Z the proton number[edit]
All consideration of nuclear electrons ended with James Chadwick's discovery of the neutron in 1932. An atom of gold now was seen as containing 118 neutrons rather than 118 nuclear electrons, and its positive charge now was realized to come entirely from a content of 79 protons. After 1932, therefore, an element's atomic number Z was also realized to be identical to the proton number of its nuclei.
The symbol of Z[edit]
Atomic Number Mass Number Element
The conventional symbol Z possibly comes from the German word Atomzahl (atomic number).[8] However, prior to 1915, the word Zahl (simply number) was used for an element's assigned number in the periodic table.
Chemical properties[edit]
Each element has a specific set of chemical properties as a consequence of the number of electrons present in the neutral atom, which is Z (the atomic number). The configuration of these electrons follows from the principles of quantum mechanics. The number of electrons in each element's electron shells, particularly the outermost valence shell, is the primary factor in determining its chemical bonding behavior. Hence, it is the atomic number alone that determines the chemical properties of an element; and it is for this reason that an element can be defined as consisting of any mixture of atoms with a given atomic number.
New elements[edit]
The quest for new elements is usually described using atomic numbers. As of 2021, all elements with atomic numbers 1 to 118 have been observed. Synthesis of new elements is accomplished by bombarding target atoms of heavy elements with ions, such that the sum of the atomic numbers of the target and ion elements equals the atomic number of the element being created. In general, the half-life of a nuclide becomes shorter as atomic number increases, though undiscovered nuclides with certain 'magic' numbers of protons and neutrons may have relatively longer half-lives and comprise an island of stability.
See also[edit]
| Look up atomic number in Wiktionary, the free dictionary. |
References[edit]


- ^ abThe Periodic Table of Elements, American Institute of Physics
- ^The Development of the Periodic Table, Royal Society of Chemistry
- ^Ordering the Elements in the Periodic Table, Royal Chemical Society
- ^Moseley, H.G.J. (1913). 'XCIII.The high-frequency spectra of the elements'. Philosophical Magazine. Series 6. 26 (156): 1024. doi:10.1080/14786441308635052. Archived from the original on 22 January 2010.
- ^Eric Scerri, A tale of seven elements, (Oxford University Press 2013) ISBN978-0-19-539131-2, p.47
- ^Scerri chaps. 3–9 (one chapter per element)
- ^Ernest Rutherford | NZHistory.net.nz, New Zealand history online. Nzhistory.net.nz (19 October 1937). Retrieved on 2011-01-26.
- ^Origin of symbol Z. frostburg.edu
